Description
Overview
The TriTom imaging platform utilizes photoacoustic tomographies (PAFT) to enable high-resolution (up to 150 μm) non-invasive in vivo whole-body imaging in small animal models. With the ability to use multiple excitation wavelengths per scan, the multi-modality system can simultaneously acquire photoacoustic data in large volumes (30-60 cm3) allowing for spectroscopic molecular analysis within the region of interest. In addition to 3D molecular maps, the TriTom enables spatially-resolved assessment of physiologic parameters in vivo, such as volumetric blood content and oxygenation without the need for contrast agents. With such versatile imaging capabilities, the state-of-the-art TriTom system is ideally suited for a wide range of preclinical applications including cancer, toxicology, developmental biology, tissue engineering and regeneration, neuroscience, cardiovascular imaging, as well as in the development of drugs, therapies, and optical and fluorescent imaging probes.
The TriTom is specially designed for imaging small animal models with a convenient animal handling set-up for fast, reproducible positioning of the subject and integrated gas anesthesia to quickly move from animal preparation to imaging. The rapid image acquisition time (~36 seconds) allows for fast imaging of individual subjects or capturing dynamic processes such as biodistribution, lymphatic drainage, or vasodilation. Additionally, the temperature control unit maintains the imaging environment within ± 0.1°C, ensuring the animal’s welfare throughout the imaging session.
A user-friendly image acquisition software enables new users to start collecting data quickly while maintaining the flexibility for advanced users to optimize imaging parameters for their specific research applications. The image reconstruction software is also designed for rapid reconstruction and visualization of large volumes with an open data format that allows for flexibility in image reconstruction and data management. With such versatile imaging capabilities, the state-of-the-art TriTom system is ideally suited for a wide range of preclinical applications.
Features
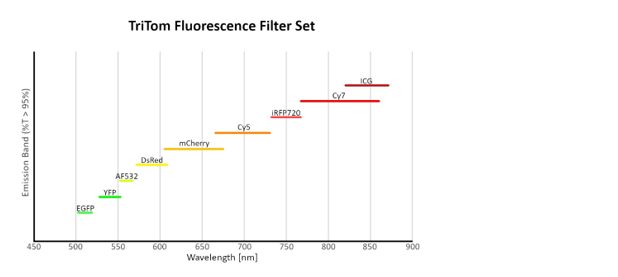
PA excitation wavelength range 532nm & 650-1320nm, covering visible light, NIR I and NIR II excitation light sources
3D photoacoustic tomography in a compact benchtop design
Fast imaging scans (<36 s) of large volumes (30-60 cm3) with superior molecular sensitivity
Accurate 3D registration of anatomical, functional, and molecular volumes with a high spatial resolution (up to 150 μm)
Tunable optical excitation wavelength across the entire spectral range, enabling multiple wavelengths in a single scan
Integrated gas anesthesia line and adjustable mouse holder for convenient operation and repeatable in vivo longitudinal studies
Single-scan assessment of multiple micro samples (50 μL or less) to accelerate contrast agent development without wasting expensive samples
User-friendly integrated software suite designed to minimize experiment time and reconstruct large-scale volumes in seconds
Open data format allowing for image reconstruction and data management with third-party software

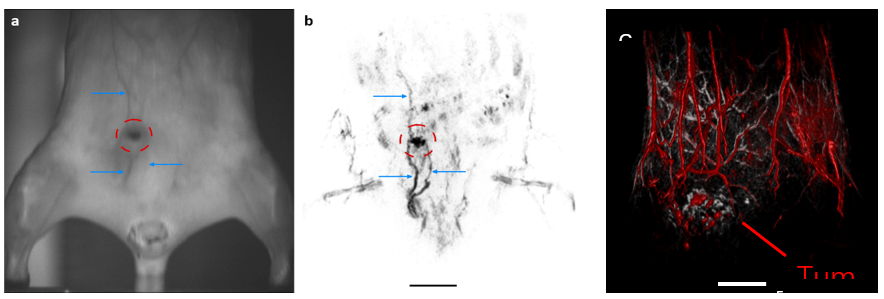
Human breast ductal carcinoma xenograft (BT474 cells) in a hairless (left) and nu/nu mouse (right).(a, b) show the camera observation and corresponding coronal MIP slab constructed from TriTom images acquired with 700-nm and 1064-nm laser excitation. The blue arrows and red ROI indicate the superficial vascular features and tumor environment visible in the high-resolution images. (c) Composite skin (532 nm; grey) and deep-tissue (890 nm; red) 3D images. Tumor size = 10.6 x 4.7 x 11.6 mm3
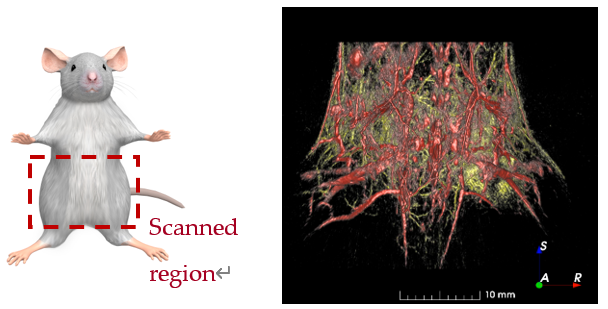
TriTom composite image of deep and superficial blood vessels in a live mouse model of metastatic breast cancer
Red – Deep vessel(890 nm PA excitation)
Yellow – Skin Level Vessel(532 nm PA excitation)

ICG & Photoacoustic molecular dissociation imaging of hemoglobin

Mouse vascular imaging without any Contrast agents
Contrast agents development
Journal Citations
I. B. Belyaev et al., "Laser-Synthesized Germanium Nanoparticles as Biodearadable Material for Near.Infrared Photoacoustic lmaging and Cancer Phototherapy," AdvSci 2307060, 2024, doi: 10.1002/advs.202307060.
R. M. Cam, C. Wang, w. Thompson, S. A. Ermilov, M. A. Anastasio, and u. Villa, "Spatiotemporal lmageReconstruction to Enable High-Frame Rate Dynamic Photoacoustic Tomography with Rotating-Gantry Volumetric lmagers," ArXiv, 2023, doi: 10.48550/arXiv.2310.00529.
K. Huda, D. J. Lawrence, w. Thompson, S. H. Lindsey, and C. L. Bayer, "in vivo noninvasive systemicmyography of acute systemic vasoactivity in female pregnant mice," Nature Communications, vol14,no.1,2023,doi: 10.1038/s41467-023-42041-8.
V. D. Vincely and C. L. Bayer, "Functional photoacoustic imaging for placental monitoring: A minireview," lEEE Transactions on Ultrasonics, Ferroelectrics, and Frequency Control, pp. 1-1, 2023, doi.10.1109/tuffc.2023.3263361.
W. R. Thompson et al., "Characterizing a photoacoustic and fluorescence imaging platform forpreclinical murine longitudinal studies," 1. Biomed. Opt, vol. 28, no. 3, p. 036001, 2023, doi:10.1117/1.JBO.28.3.036001.
M. Delcroix, A. Reddy Marri, S. Parant, P. C. Gros, and M. Bouché, "Water-soluble Fe(l) complexes fortheranostic application: Synthesis, photoacoustic imaging and photothermal conversion," Eur. ..Inorg.Chem,, vol.26, no.27,2023, doi: 10.1002/eiic.202300138.
S. Singh et al., "Size-tunable lCG-based contrast agent platform for targeted near-infraredphotoacoustic imaging," Photoacoustics, vol. 29, p.100437, 2023, doi: 10.1016/i.pacs.2022.100437
Z. Zhao, c. B. Swartchick, and J. Chan, "Targeted contrast agents and activatable probes forphotoacoustic imaging of cancer," Chem Soc Rev, vol. 5l, no.3, pp. 829-868, 2022, doi.10.1039/d0cs00771d.
J. Kim, A. M. Yu, K. P. Kubelick, and S. Y. Emelianov, "Gold nanoparticles conjugated with DNAaptamer for photoacoustic detection of human matrix metalloproteinase-9," Photoacoustics, vol. 25p.100307, 2022, doi: 10.1016/i.pacs.2021.100307.
M. R. Chetyrkina et al., "Carbon Nanotube Microscale Fiber Grid as an Advanced Calibration Systemfor Multispectral Optoacoustic lmaging," Acs Photonics, vol. 9, no.10, pp. 3429-3439, 2022, doi:10.1021/acsphotonics.2c01074.
M. D. Mokrousov et al, "indocyanine green dye based bimodal contrast agent tested byphotoacoustic/fluorescence tomography setup," Biomed. Opt. Express, vol. 12, no. 6, p. 3181, 2021, doi.10.1364/boe.419461.
A. Juronis and M. Jasinskas, "Breakthrough instruments and products PhotoSonus M+ laser forphotoacoustic imaging," Review of Scientific instruments, vol. 92, no. 5, p. 059502, 2021, doi.10.1063/5.0053559.
K. Huda, c. wu, J. G. Sider, and c. L, Baver, "Spherical-view photoacoustic tomography for monitoringin vivo placental function," Photoacoustics, vol. 20,p.100209, 2020, doi: 10.1016/i.pacs.2020.100209
E. M. Donnelly, K. P. Kubelick, D.s. Dumani, and S. Y. Emelianov, "Photoacoustic lmage-GuidedDelivery of Plasmonic-Nanoparticle-Labeled Mesenchymal Stem Cells to the Spinal Cord," NanoLetters, vol.18, no.10, pp.6625-6632,2018, doi: 10.1021/acs.nanolett.8b03305


